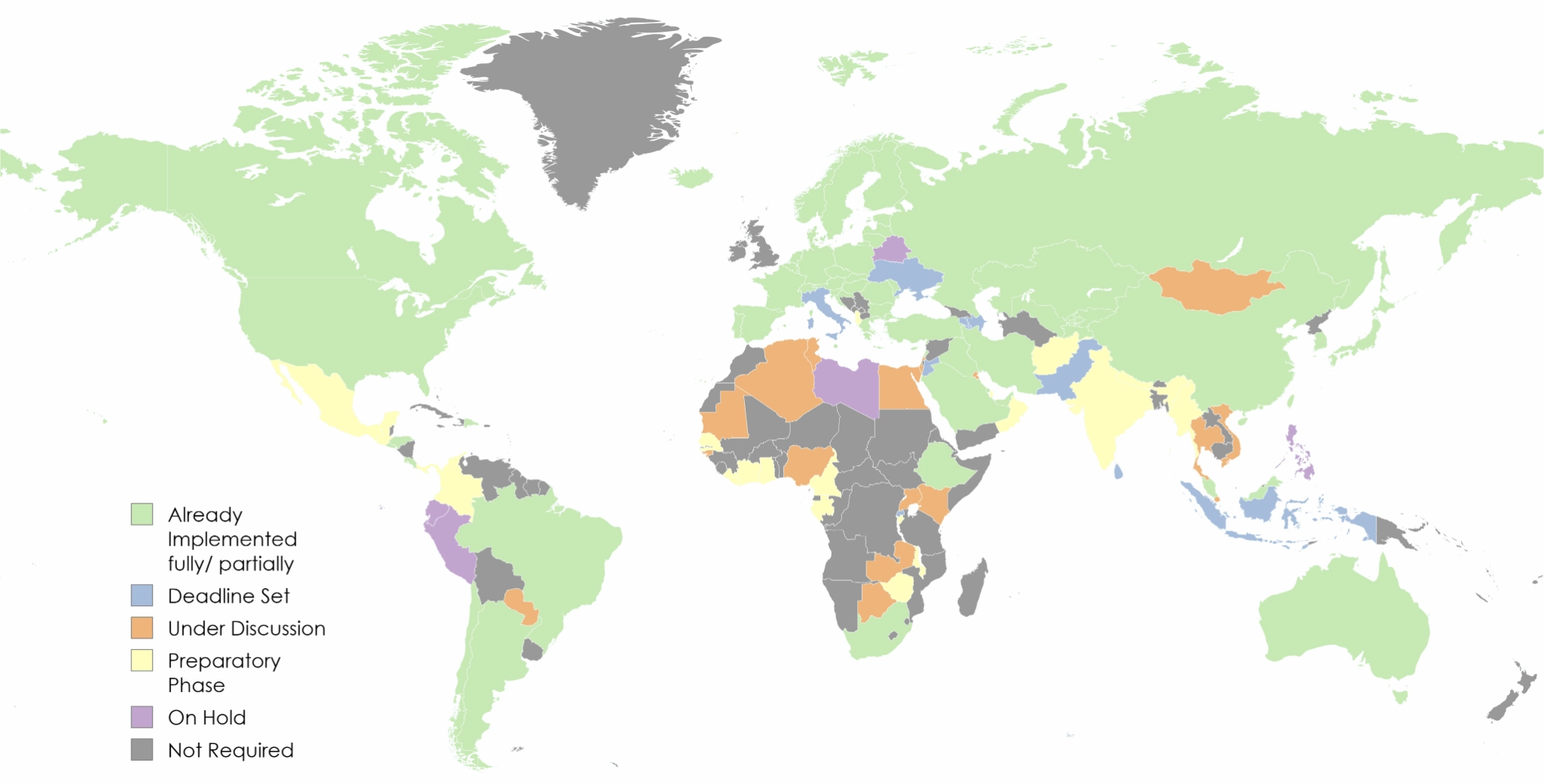
A live, country-by-country view of where pharmaceutical serialization and traceability is mandatory, where deadlines are approaching, and where regulation is still under discussion. Updated continuously by SoftGroup’s regulatory team based on official government and authority sources.
How to read this tracker
We group countries into five regulatory states. Click any country to expand details, including effective date, governing regulation, and the national hub or gateway in use.
- Already Implemented — Regulation is in force; compliance is mandatory for sale or distribution.
- Deadline Set — Regulation is published; enforcement begins on a fixed future date.
- Under Discussion — Draft regulation, public consultation, or pilot underway. No binding date.
- On Hold — Originally announced or expected; currently paused, deferred, or withdrawn.
- Not Required — No national serialization mandate at present. Voluntary GS1 standards may still apply.
Methodology note : Statuses are reviewed at least monthly against primary sources — Ministry of Health publications, regulatory gazettes, national hub announcements, GS1 country chapters, and direct dialogue with our partner network. If you spot something outdated, tell us.
Already Implemented — Mandatory serialization in force
Twenty-six markets currently require serialization, aggregation, or full track-and-trace for some or all pharmaceutical products. The European Union, United States, and most major Asian and Middle East markets fall in this category.
| Country | In Force Since | Framework / Hub |
|---|---|---|
| European Union (27 + EEA) | 9 February 2019 | EU FMD via EMVS / EMVO |
| Greece | 9 February 2025 | National deferral ended; full NMVS now active |
| Belgium | 9 February 2025 | National deferral ended; full NMVS now active |
| Turkey | January 2010 | İTS (Turkish Pharmaceuticals Track & Trace System) |
| Country | In Force Since | Framework |
|---|---|---|
| Argentina | 2015 | ANMAT Trazabilidad |
| Brazil | April 2022 | ANVISA SNCM |
| Canada | December 2021 | Health Canada GUI-0001 / DPD |
| United States | 27 May 2025 | DSCSA — full enforcement after stabilization period |
| Country | In Force Since | Framework |
|---|---|---|
| Australia | 2024 | TGA recall and supply chain regulations |
| China | 2022 | NMPA drug traceability code system |
| India | January 2023 (export) | DGFT — exported formulations; iVEDA portal |
| Japan | January 2023 | MHLW barcode labeling, full implementation |
| South Korea | January 2015 | KGMP serialization, KPIS reporting |
| Country | In Force Since | Framework |
|---|---|---|
| Bahrain | 2022 | NHRA-MVC (market expansion details) |
| Ethiopia | February 2025 | EFDA traceability regulation |
| Iran | January 2015 | TTAC |
| Lebanon | January 2023 | MoPH |
| Oman | March 2019 | MoH serialization |
| Saudi Arabia | March 2017 | SFDA RSD |
| South Africa | June 2022 | SAHPRA labelling regulation |
| UAE | January 2022 | Tatmeen platform |
| Country | In Force Since | Framework / Hub |
|---|---|---|
| Kazakhstan | July 2024 | NaMSED / EBD (market expansion details) |
| Kyrgyzstan | 2023–2024 (phased) | ЦДМ / EBD |
| Russia | July 2020 | Chestny ZNAK / CRPT |
| Tajikistan | January 2025 | National traceability system |
| Uzbekistan | February 2022 | ASL Belgisi |
SoftGroup connects MAHs and manufacturers to all of the national hubs above through the SATT Platform. See how multi-market integration works
Deadline Set — Regulation published, enforcement coming
| Country | Enforcement Date | Notes |
|---|---|---|
| Indonesia | December 2025 | BPOM 2D barcode mandate — transition window in progress |
| Sri Lanka | December 2025 | NMRA serialization rule — transition window in progress |
| Armenia | January 2026 | Phased rollout via national MDLP equivalent |
| Italy | February 2027 | National derogation under EU FMD ends; full NMVS go-live |
| Rwanda | August 2026 | FDA Rwanda national traceability system |
| Malaysia | 2027 | NPRA — final scope under finalization |
| Pakistan | TBA — published, awaiting calendar | DRAP serialization regulation |
| Ukraine | January 2028 | Postponed twice; current target subject to security situation |
Indonesia and Sri Lanka are now within their official transition windows — practical enforcement is being applied with discretion through the first half of 2026. MAHs supplying either market should treat go-live as now, not December 2025.
Under Discussion — Draft regulation or pilot phase
Fourteen markets have an active serialization or traceability initiative without binding enforcement dates. The list moves quickly, we re-check every status monthly.
| Country | Status | Indicative Timeline |
|---|---|---|
| Algeria | Draft regulation under MoH | TBA |
| Azerbaijan | DVTIS platform piloted | Originally January 2026 — under review |
| Botswana | National policy drafted | Targeting 2027 |
| Egypt | EDA traceability roadmap | Multi-year rollout |
| Georgia | Working group active | TBA |
| Jordan | JFDA pilot extended | Originally June 2025 — extended |
| Kuwait | MoH consultation | 2026 / 2027 |
| Kenya | PPB drafting framework | TBA |
| Nigeria | NAFDAC MAS evolving toward full serialization | TBA |
| Qatar | MoPH consultation | TBA |
| Tunisia | DPM working group | 2027 / 2028 |
| Uganda | NDA initial framework | 2027 / 2029 |
| Vietnam | DAV pilot programs | TBA |
| Zambia | ZAMRA draft regulation | Targeting 2028 |
On Hold — Announced or expected but currently paused
Six markets previously announced serialization regulation that is now postponed, withdrawn, or stalled without an active timeline.
| Country | Status |
|---|---|
| Belarus | Serialization framework drafted; politically paused |
| Ecuador | Initial regulation; implementation deferred |
| Libya | Framework discussion; on hold pending broader regulatory reform |
| Paraguay | Initial draft; no active calendar |
| Peru | Initial framework; DIGEMID activity paused |
| Philippines | FDA Philippines draft circular; no binding calendar |
“On hold” is not “never.” Re-activation typically happens with little public warning — usually tied to a specific government priority or a regional trade alignment. Monitor status quarterly if you sell into any of these markets.
Not Required — No national mandate at present
A growing list of markets have no national serialization mandate but may still require:
- GS1-compliant barcodes for reimbursement, hospital tendering, or e-procurement
- Voluntary aggregation for exports into mandated markets
- Cold-chain traceability for biologics, even where it isn’t framed as “serialization”
Examples include most of sub-Saharan Africa outside the markets listed above, several Pacific island nations, and a number of Caribbean and Central American countries.
Operational reality: If you export from a “Not Required” country into a regulated market, the regulation of the destination still applies. Serialization is a destination-driven obligation in nearly all frameworks.
How SoftGroup helps in every category
| Your situation | What we deliver |
|---|---|
| Operating in a market that's Already Implemented | Direct connection from your line to the national hub via the SATT Platform — single integration, validated, audit-ready. |
| Preparing for a market with a Deadline Set | Pre-built connectors for most national hubs, validated GxP templates, and predictable cutover plans. |
| Watching a market Under Discussion | Country intelligence and pre-engagement so you're not learning the regulation after it's enforceable. |
| Operating across multiple categories at once | Single architecture, one MAH view, no parallel vendor stacks. Explore multi-market → |
| Switching from a vendor that can't keep up | Vendor switch playbook → |
| Coordinating partner CMOs across markets | CMO serialization platform → |
Frequently asked questions
Which countries currently require pharmaceutical serialization?
As of May 2026, 26 markets have mandatory serialization in force, including the European Union (under EU FMD), the United States (under DSCSA, fully enforceable from 27 May 2025), Russia, China, India, Japan, South Korea, Turkey, Saudi Arabia, UAE, and most CIS states. A further 8 markets have a binding deadline set, and 14 more are in active consultation.
Is global pharmaceutical serialization the same as track and trace?
They overlap but aren’t identical. Serialization is the act of applying a unique identifier to each saleable pack. Track and trace adds the requirement to record movements of that pack through the supply chain. Most modern regulations — EU FMD, US DSCSA, Russia CRPT, UAE Tatmeen — require both serialization and downstream verification or trace.
What's the difference between EU FMD and other national systems?
U FMD is a point-of-dispense verification model — packs are verified when dispensed by a pharmacy. Many non-EU systems are full traceability models — every movement is reported to a national hub. Both require serialization at the manufacturing line; the downstream obligations differ.
Do MAHs need a different solution per country?
ot necessarily. A platform like SoftGroup’s SATT PLATFORM provides a single integration layer that connects to most national hubs (EMVO, Tatmeen, ASL Belgisi, CRPT, Chestny ZNAK, EBD, DVTIS, NHRA-MVC, and more). The alternative — running parallel vendor stacks per market — is operationally fragile and expensive at scale.
Need country-specific compliance intelligence? Tell us which markets you’re operating in or expanding into. We’ll respond within one business day with a country-by-country compliance brief and an architecture proposal that covers all of them from a single platform.